
Protein Delivery into Plant Cells by Electroporation
In plant genetic engineering and biotechnology, the effective delivery of biomolecules such as proteins into plant cells remains a significant challenge. Particularly in plant cells with rigid cell walls, the penetration of large molecules is problematic. Conventional gene transfection methods, such as those using Agrobacterium or the gene gun, are employed to introduce DNA into these cells; however, the entry of foreign DNA and its integration into the host genome can lead to unintended consequences.
In contrast, direct protein delivery, which eliminates the need for DNA, can circumvent many of these issues. Plant cell electroporation is an effective physical method that temporarily permeabilizes the cell membrane by applying short electrical pulses, thereby enabling the delivery of proteins into the cell. In recent years, advancements in equipment and methods have led to an expansion in the use of electroporation in plant laboratories. Additionally, numerous studies have been conducted on the use of electroporation in algae. Below, the principles of electroporation and its application in protein delivery to plant cells will be examined.
.
Electroporation: Definitions and Applications
Electroporation is a method that enables the delivery of large molecules, such as DNA, RNA, and proteins, into cells by creating temporary pores in the cell membrane through the application of an electric field. In other words, short, high-intensity electrical pulses cause temporary openings in the cell membrane, allowing target molecules to penetrate into the cytoplasm through these channels.
Electroporation cuvettes contain metal electrodes and safety caps that hold the solution containing cells or proteins, allowing the electrical pulse to pass through them. In wall-less cells, such as those of animals or yeast, electroporation is a standard method for delivering DNA and proteins due to its simplicity and high efficiency. However, in plants, the presence of the cell wall complicates this process.
.
Challenges and Conventional Methods for Delivering Molecules into Plant Cells
Plant cells possess a cell wall that makes the penetration of biological molecules into the cell much more difficult. Therefore, one strategy to overcome this barrier is to convert plant cells into protoplasts through the enzymatic removal of the cell wall. Studies have shown that protoplasts from plants such as tobacco and Arabidopsis can receive DNA, RNA, and even proteins; however, regenerating whole plants from protoplasts is either impossible or inefficient in many species.
On the other hand, other physical methods such as particle bombardment (gene gun) or delivery via Agrobacterium (Rhizobium) are also standard for introducing genetic material into plant cells. Although these methods enable efficient expression of engineered genes, the deposited extra DNA often integrates into the host plant genome and can cause unintended side effects. For this reason, methods that can deliver proteins directly into plant cells without involving foreign DNA have gained increasing interest.
.
Electroporation in Protein Delivery to Plant Cells
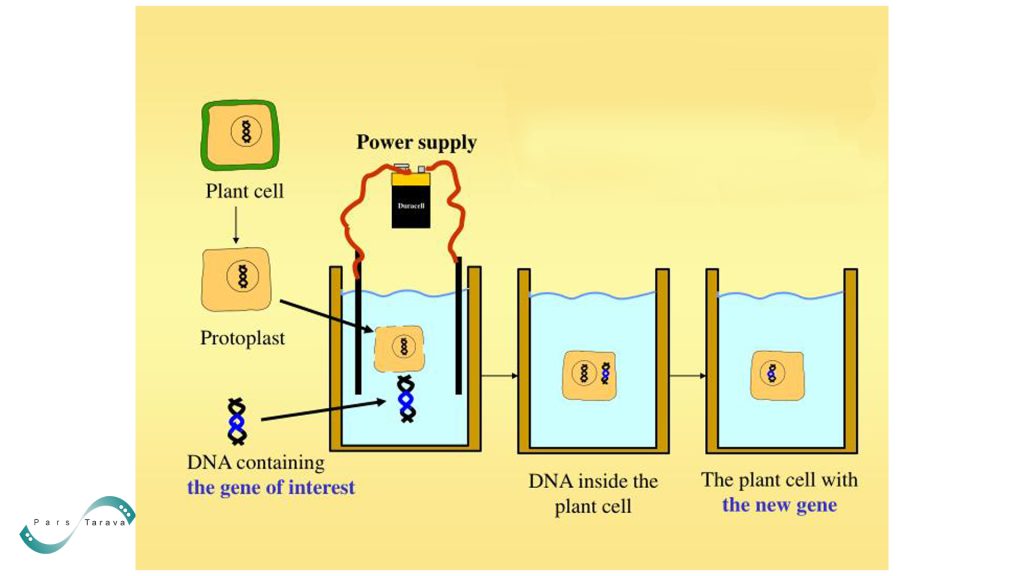
Numerous studies have demonstrated that plant cell electroporation can facilitate the successful delivery of proteins into plant cells with cell walls. For example, one of the most famous genetic engineering tools is a system called Cre/loxP. The use of this system in plants is more limited. One primary reason for this limitation is the presence of a rigid wall around plant cells, which makes delivering required materials (such as Cre enzyme or instructions for its production) into the cell difficult.
In a study, Furuhata et al. (2019) successfully delivered Cre recombinase protein into over 80% of Arabidopsis cells by applying multiple electrical pulses. They used Opti-MEM I medium for electroporation and reported that this medium offers higher delivery efficiency and lower toxicity compared to conventional buffers.
In this research, five pulses of 375 V/cm for 10 milliseconds were typically used, resulting in approximately 83% delivery efficiency. Furthermore, it was determined that a minimum concentration of about 0.2 micromolar of Cre protein is necessary to observe effective delivery. These results indicate that by optimizing electroporation parameters, very high efficiency in delivering proteins into plant cells can be achieved.
.
Key Points to Increase Protein Electroporation Efficiency:
- Electroporation buffer: Selecting an appropriate medium such as Opti-MEM I to prevent cellular toxicity and increase delivery efficiency.
- Pulse parameters: Precise adjustment of voltage intensity (several hundred V/cm), duration, and number of pulses to balance delivery and cell viability (e.g., 375 V/cm for 10 ms).
- Protein concentration: Using sufficient concentration (above approximately 0.2 µM for Cre recombinase) to achieve high efficiency.
- Cell density: The state of cells (single or clustered) and their density can impact the effectiveness of protein delivery; in some studies, the dissociation and isolation of clustered cells have shown improved efficiency.
.
Examples of Successful Protein Delivery
In a recent study, Carrillo-Carrasco et al. (2023) successfully delivered proteins into the cells of a green alga named Penium margaritaceum using electroporation, without the need for DNA involvement. They identified optimal conditions that maintained high delivery efficiency and cell viability. Additionally, a novel method called DIVE (Device-Independent protein VectEring) has been introduced, achieving approximately 94% efficiency in Arabidopsis cells by placing cells in a protein-containing medium without the need for an electrical pulse. These results suggest that innovative methods for delivering proteins into plant cells are emerging.
.
Electroporation Equipment
Pars Tarava Company is the first and sole manufacturer of electroporation devices in Iran. Additionally, by designing and constructing various electrodes tailored for plant research, this company has paved the way for further research and studies on plants. Dear researchers, you can contact 00989024051862 for a free consultation and to order electroporation devices, including 2- and 4-mm electroporation cuvettes, as well as other electrodes.
.
Challenges and Limitations
- Cellular toxicity: Applying intense electrical pulses can damage and kill plant cells; therefore, precise adjustment of voltage and pulse duration is critical for preserving cell viability.
- Inter-species differences: Cell wall thickness and composition vary among different plant species. Consequently, optimal electroporation conditions (such as voltage and pulse duration) may differ for each plant species.
- Importance of suitable equipment: Successful electroporation requires specialized devices and controlled environments. Technological development and domestic equipment production (such as efforts by Pars Tarava Company) can help address these limitations.
.
Conclusion
Recent studies have demonstrated that by optimizing electrical conditions and delivery media, high efficiency (over 80%) in protein delivery to plant cells can be achieved. On the other hand, direct protein delivery via this method eliminates the risks of random integration of extra DNA into the genome. Furthermore, delivering proteins instead of DNA enables faster genetic experiments and manipulations. Applications of this method in protein structure-function studies within the cellular environment, investigation of signaling pathways, and production of therapeutic molecules in plants are extensive.
Pars Tarava Company is the first manufacturer of plant cell electroporation devices in Iran, designing and producing various electroporation equipment. For more information and specialized consultation regarding electroporation products, you can contact 00989024051862.
.
Sources
Gene transfer to plants by electroporation: methods and applications
Electroporation-based delivery of proteins in Penium margaritaceum and other zygnematophycean algae
Direct protein delivery into intact Arabidopsis cells for genome engineering
.
